PRESS & NEWS
Quick Question March 2023 Results: Which FTD Registry communications help you learn about reach opportunities?

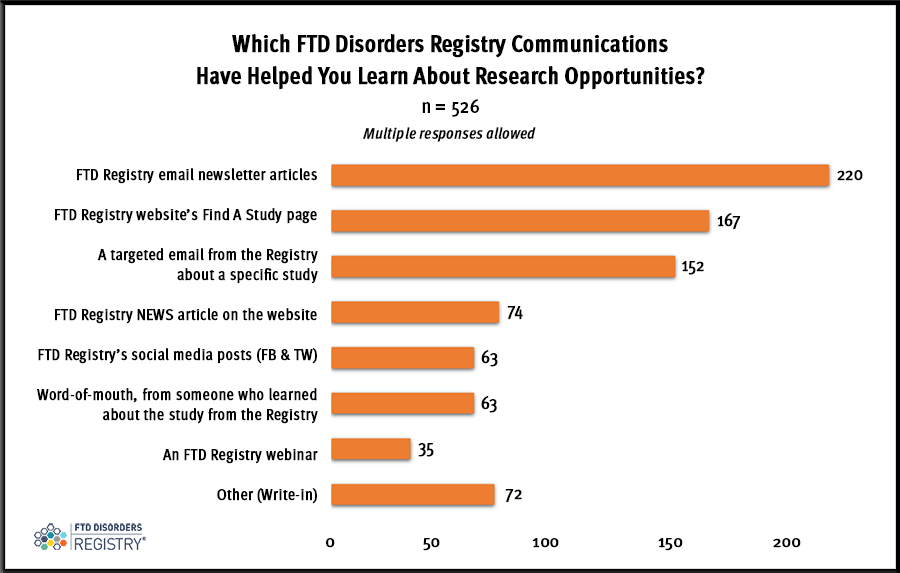
The FTD Registry’s email newsletter articles are the No. 1 way people learn about FTD research studies through the FTD Disorders Registry, according to the March 2023 Quick Question.
In March 2023, there were 526 people who reported on the different types of FTD Disorders Registry communication methods they use to learn about research studies for frontotemporal degeneration (FTD). The greatest response (220) was the Registry’s email newsletter articles.
Checking listings on the FTD Registry’s Find A Study webpage earned the second most responses (167), followed closely by Registry emails that refer to a specific study (152). Topical emails are sent to people who have signed up and consented to research. Email lists are created based on responses to the Registry’s three core surveys.
Most of the write-in responses related to non-Registry channels, some of these included physicians, clinicians, and other medical staff (16); universities (11); and the Association for Frontotemporal Degeneration (7).
Replies from 10 people who submitted write-in answers were related to not being able to participate in research, including not knowing of any studies, having no studies nearby, diagnosed loved one passed before there was a Registry, and being too new to the Registry.
Of the 526 responses, 94 came from people diagnosed with FTD. Biological family members (137), spouses (82), caregivers (14), and friends (2) answered for themselves. They were also invited to complete the poll on behalf of their FTD-diagnosed loved one [spouses (139), biological family members (43), caregivers (10), and friends (1)]. Four people did not identify their relationship.
Most people answering this month’s question were from 45 U.S. states (473) and the District of Columbia (2). There were 33 people from 9 Canadian provinces and territories who replied. Australia (6) and the United Kingdom of Great Britain and Northern Ireland (5) complete the top three countries with the highest number of respondents.
RESULTS
Each month the FTD Disorders Registry asks a question seeking feedback. Answers are anonymous.
Multiple responses were allowed in March so that everyone could include all of the ways they seek information about research studies from FTD Registry sources..
RELATIONSHIP TO FTD-DIAGNOSED LOVED ONE
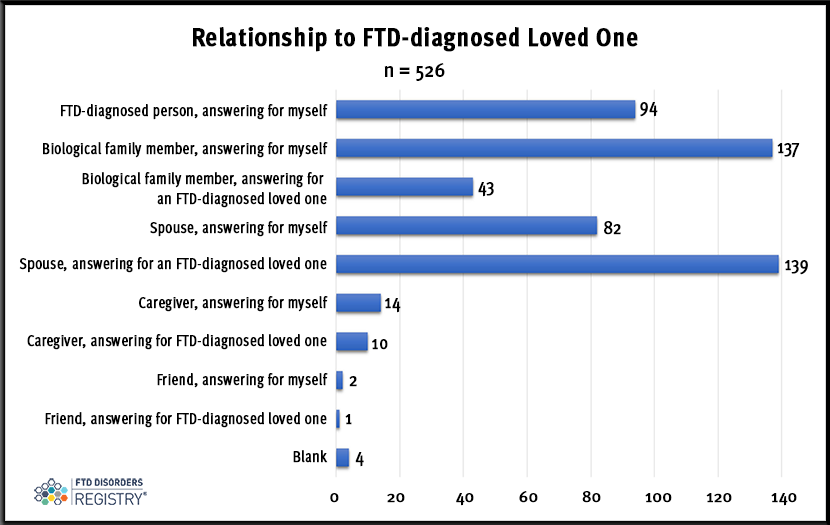
Spouses provided the greatest number of answers with 221. There were 82 who answered for themselves and 139 answered on behalf of their FTD-diagnosed loved one.
Some 94 persons diagnosed with FTD responded to the question. Spouses, biological family members, caregivers, and friends could answer for themselves and then complete the poll again on behalf of their FTD-diagnosed loved one.
Relationship to the FTD-diagnosed person:
You can access previous questions and responses on the Quick Questions summary page.
Together we can find a cure for ftd
The FTD Disorders Registry is a powerful tool in the movement to create therapies and find a cure. Together we can help change the course of the disease and put an end to FTD.
Your privacy is important! We promise to protect it. We will not share your contact information.



